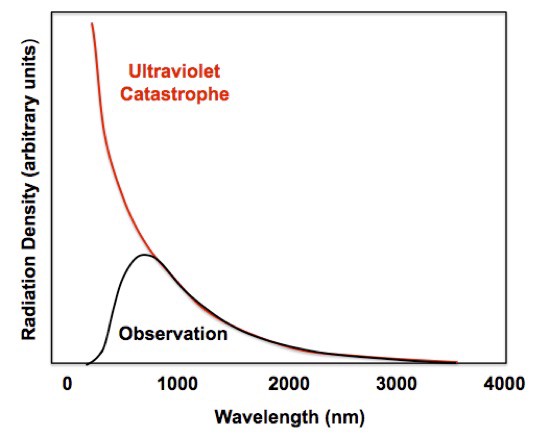
While we discussed in class the importance of blackbody spectra continuous spectra, there is an important historical footnote in understanding where the famous blackbody curve arises from. Physics in the late 1800’s and early 1900’s predicted the wavelength-intensity relationship to be I α 1/λ^4, using a derivation based on classical statistical mechanics. This relationship closely matches the observed blackbody electromagnetic radiation for high wavelengths, but is not accurate for lower wavelengths, particularly diverging in the ultraviolet spectrum. This divergence, that intensity ought to be going to infinity as wavelength goes to zero, came to be known as the ultraviolet catastrophe. The solution to this catastrophe was formulated in the early 1900’s by Max Planck. He started from the assumption that there was a minimal amount that energy could change by while emitting blackbody radiation. With this assumption, he was able to match the observed data far more accurately, creating Planck’s law based on strictly empirical data.
This assumption of a minimal energy change became the foundation of quantum mechanics. Planck’s insight that energy was quantized was later justified by Einstein through his analysis of the photoelectric effect. This empirical guess that the relationship could match a quantized output level turned out to be a precursor of the existence of photons. The way the blackbody curve looks is fundamentally due to quantum mechanics, and the historical importance of it is hard to overstate.

So important, and the addition of contaminants within the star’s atmosphere play such a big role in examining a blackbody curve
LikeLike